Get the full story of the microbiome
- Importance of Microbial Diversity
- Dysbiosis and Recurrent C. difficile Infection
- Historic Microbiome Restoration Approaches
- Patient and Healthcare Burdens
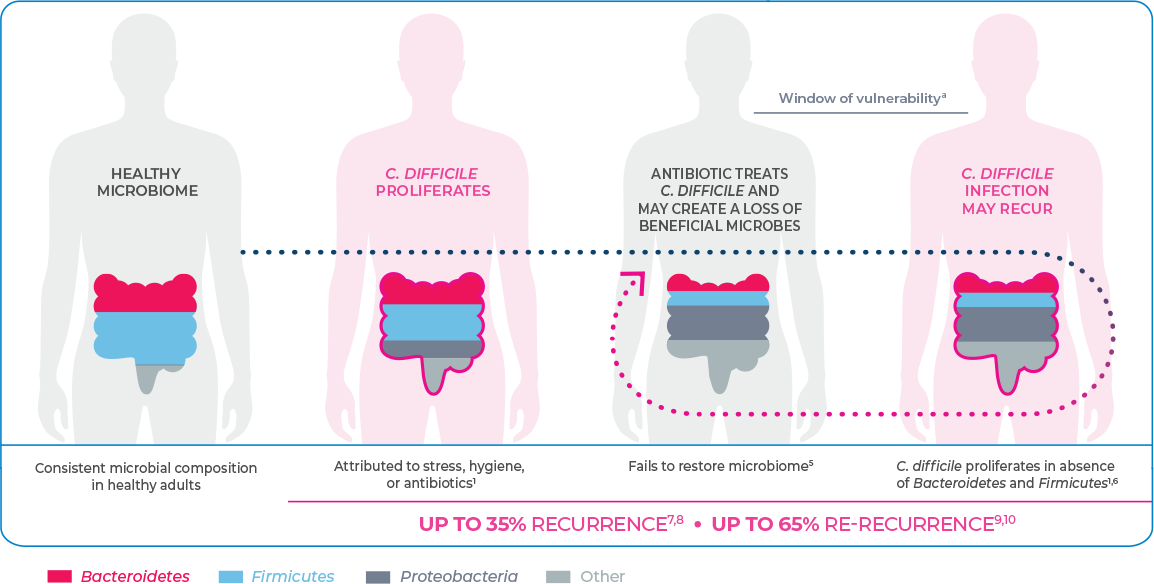
C. difficile may proliferate as a result of dysbiosis
Dysbiosis creates an insufficiency of Bacteroidetes and Firmicutes, which may lead to an environment suited for the growth of C. difficile infection (CDI).1,2
Dysbiosis is the disruption of the volume and diversity of the gut microbiome. This may be attributed to factors including stress, diet, hygiene, and antibiotic use. Dysbiosis is associated with a range of gastrointestinal (GI) and non-GI diseases including neurologic, metabolic, liver, inflammatory, and infectious diseases.1,3,4
The gut microbiota generally play a role in colonization resistance by which the native organisms prevent pathogenic microbes from flourishing. Disruption of the gut microbiome leads to an environment suited for the proliferation of C. difficile.1,2
THE BURDEN OF CDI MAY INCREASE
WHEN THE INFECTION RECURS
Can the power of the microbiome be
unlocked to break the cycle of rCDI?
Share this with colleagues!

References
- Bien J, Palagani V, Bozko P. The intestinal microbiota dysbiosis and Clostridium difficile infection: is there a relationship with inflammatory bowel disease? Therap Adv Gastroenterol. 2013;6(1):53-68.
- Staley C, Khoruts A, Sadowsky MJ. Contemporary applications of fecal microbiota transplantation to treat intestinal diseases in humans. Arch Med Res. 2017;48(8):766-773.
- Weiss GA, Hennet T. Mechanisms and consequences of intestinal dysbiosis. Cell Mol Life Sci. 2017;74(16):2959-2977.
- Riaz Rajoka MS, Shi J, Mehwish HM, et al. Interaction between diet composition and gut microbiota and its impact on gastrointestinal tract health. Food Science and Human Wellness. 2017;6(3):121-130.
- Langdon A, Crook N, Dantas G. The effects of antibiotics on the microbiome throughout development and alternative approaches for therapeutic modulation. Genome Med. 2016;8(1):39.
- Antharam VC, Li EC, Ishmael A, et al. Intestinal dysbiosis and depletion of butyrogenic bacteria in Clostridium difficile infection and nosocomial diarrhea. J Clin Microbiol. 2013;51(9):2884-2892.
- Lessa FC, Mu Y, Bamberg WM, et al. Burden of Clostridium difficile infection in the United States. N Engl J Med. 2015;372(9):825-834.
- Cornely OA, Miller MA, Louie TJ, Crook DW, Gorbach SL. Treatment of first recurrence of Clostridium difficile infection: fidaxomicin versus vancomycin. Clin Infect Dis. 2012;55(suppl 2):s154-s161.
- Kelly CP. Can we identify patients at high risk of recurrent Clostridium difficile infection? Clin Microbiol Infect. 2012;18(suppl 6):21-27.
- Smits WK, Lyras D, Lacy DB, Wilcox MH, Kuijper EJ. Clostridium difficile infection. Nat Rev Dis Primers. 2016;2:16020.
- Hensgens MPM, Goorhuis A, Dekkers OM, Kuijper EJ. Time interval of increased risk for Clostridium difficile infection after exposure to antibiotics. J Antimicrob Chemother. 2012;67(3):742-748.
- Wilcox MH, McGovern BH, Hecht GA. The efficacy and safety of fecal microbiota transplant for recurrent Clostridium difficile infection: current understanding and gap analysis. Open Forum Infect Dis. 2022;7(5):ofaa114.